Helicobacter heilmanii (Gastrospirillum hominis) -Bacteria
What the #&$*! is this?
While reading gastric biopsy gram preparations for Helicobacter pylori, I came across this interesting gram negative helical bacterium. It differed from the typical 'gull-winged' or wavy appearance characteristic of Helicobacter pylori by having up to eight tightly wound spirals. Roughly double the size of Helicobacter pylori, it ranges from about 3.5 to 8.0 μm in length and the spirals reach an amplitude of about 1.0 μm. In my career, this was only my third encounter with this organism, however morphology alone immediately suggested the organism most likely was Helicobacter heilmanii.
Some Background:
Helicobacter heilmanii was previously known as Gastrospirillum hominis after first being described in 1987[i]. Though figures in published literature vary, Helicobacter heilmanii appears to be responsible for less than 1% of infections of the gastric mucosa. While Helicobacter pylori is transmitted human to human, evidence suggests that Helicobacter heilmanii has an animal reservoir and is probably acquired by close association with household pets or farm animals. Children also may be prone to infection by H.heilmanii due to closer contact with family pets. This apparent 'zoonosis' also occurs more frequently in poorer socio-economic settings.
Symptoms & Pathology:
Symptoms of Helicobacter heilmanii infection vary from epigastric pain, nausea, vomiting, decreased appetite dyspepsia and chronic gastritis. Infection with H.heilmaniihas been associated with gastric and duodenal ulcers in adult patients. Cellular dysplasia, metaplasia and mucosal atrophy may possibly predispose the patient to gastric carcinoma.
Challenges in Detection and Identification:
Organisms which invade the gastric mucosa usually possess the enzyme urease which can split the urea molecule into Ammonia (NH3) and Carbon Dioxide (CO2). Tests have been developed which take advantage of this specific reaction. With the CLOtest (Campylobacter Like Organism test), a very small biopsy tissue sample is place on the CLO media and allowed to react. If organisms possessing the urease enzyme are present in the tissue, they will degrade the urea molecule as outlined above. The free CO2 dissipates however the remaining ammonia will raise the media's pH with a resulting colour change; with a negative, no colour change occurs. The alternative test is the C13 or C14“Breath Test” which utilizes urea labeled with one of the two isotopes of Carbon. (Carbon-13 is preferential as it is non-radioactive). A base-line breath level is taken after which the patient ingests a urea drink. If bacteria are present which possess the urease enzyme, the urea molecule is split into ammonia and carbon dioxide, however now the carbon dioxide is labeled with the C13 isotope. It can be distinguished from ambient carbon dioxide and the level obtained can be compared to the base line.
Drawbacks;
Sensitivity and specificity of the CLOtest varies significantly, with the product manufacturer claiming values of 98% & 97% respectively (product insert), to another evaluation having determined values of 77% & 96% respectively[ii]. Our own in-house evaluation generated even less optimistic values.
The C13 Breath Test claims 95% sensitivity and 96% specificity[iii].
Regardless, both tests rely on the bacterium's ability to metabolize the urea as well as the proper administration and interpretation of the test[iv]. It is unclear as to whether H.heilmaniiis as consistent and proficient as H.pylori in metabolizing urea and these tests cannot distinguish between the two organisms.
Serological and immunohistochemical tests for H.pyloriare available however there is cross-reactivity with H.heilmanii. As such, current antibody tests also cannot distinguish between the two species and there is no serological test specific to either.
Unlike H.pylori, H.heilmanii cannot be cultured in the routine laboratory setting and no doubt would be time consuming if a viable option.
Microscopic visualization remains the definitive test for detection and identification of Helicobacter species in biopsy specimens.
While concomitant infections with H.pylori and H.heilmaniihave been noted, they are exceedingly rare leading to unlikely speculation than one may perhaps exclude the other.
Treatment for H.heilmanii is generally accepted to be the same as for H.pylori; antibiotics and a proton pump inhibitor. I will not discuss specifics as I will leave therapy to the physicians. My intent with this post, as with all others, is simply to lay some groundwork for a few pretty pictures.
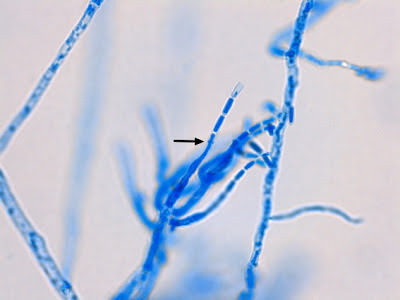
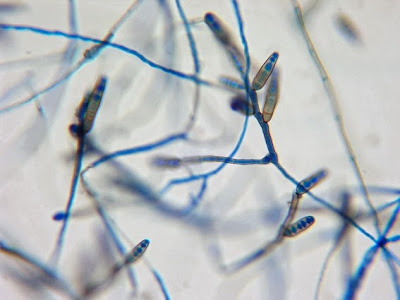
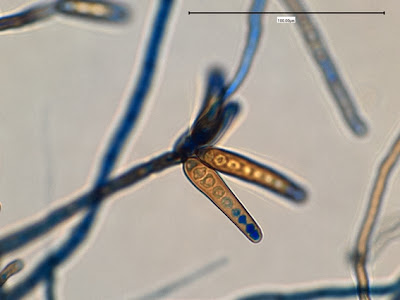
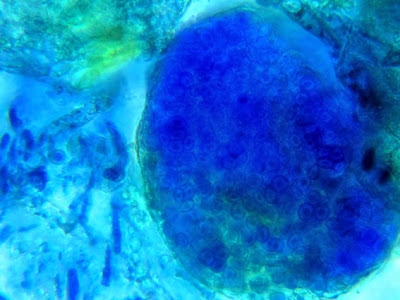
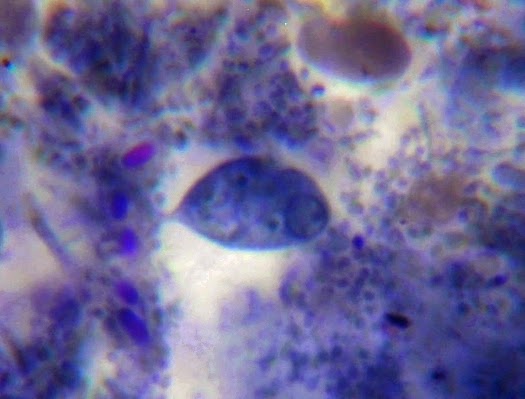
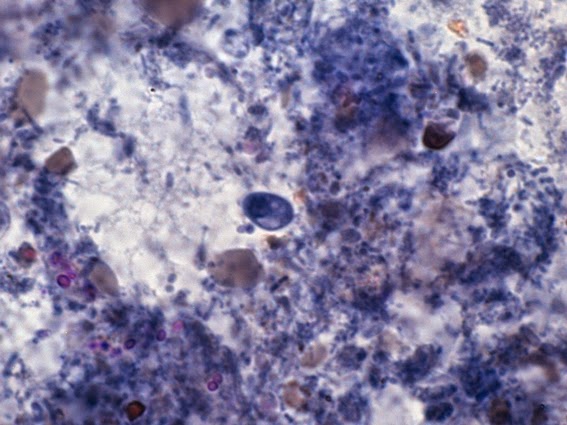
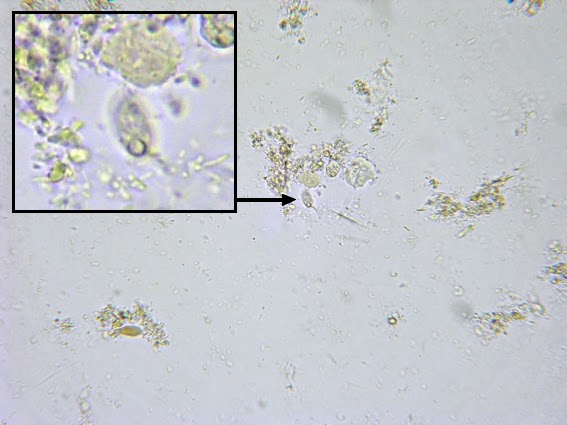
Helicobacter heilmanii - Seen here in a gastric biopsy specimen (arrows). The spiral appearance is much more evident in the enlarged insert. The other large cells are gastric mucosal cells. As we are only examining the biopsy for Helicobacter (pylori), which is a gram negative organism, there is no advantage to doing a full gram stain. A small segment of the gastric mucosa, showing evidence of ulceration, was removed during an endoscopic procedure. This tissue was mashed up onto a glass microscope slide and stained with carbol fuchsin. (carbol fuchsin appears to stain these organism more intensely than gram safranin). My experience has been that these gastric organisms seem to occur in 'pockets' in that you may search a slide for quite some time and not see a single organism, then suddenly, bang! several or even many in one area. Pass over it and nothing once again.
(Nikon, Carbol Fuchsin Stain, X1000)
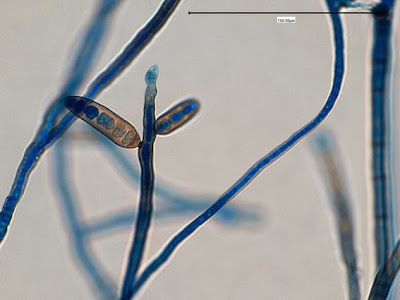
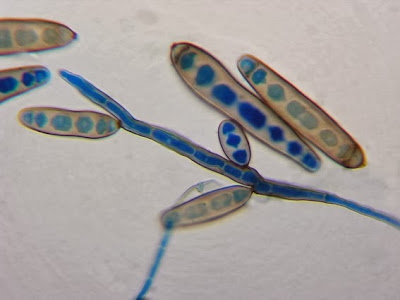
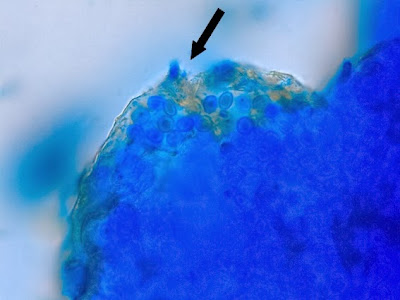
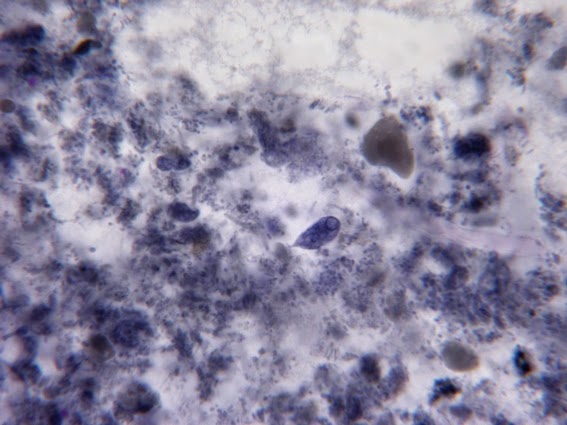
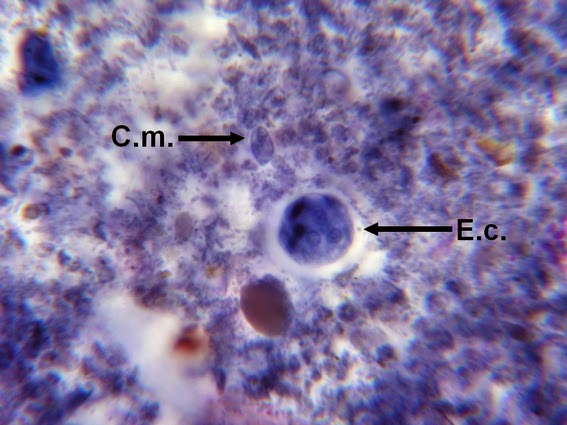
Heilicobacter heilmanii - from the same specimen as above. The resolution is better in this photo with the tight wavey spirals (upwards of 8 in number) quite evident. Organisms appear larger than above due to my cropping of the photograph.
(Nikon, Carbol Fuchsin Stain, X1000)
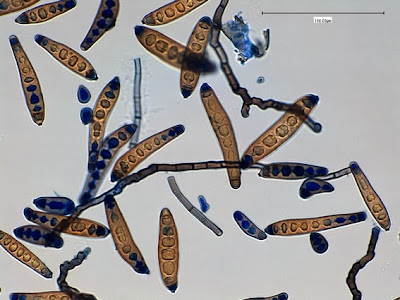
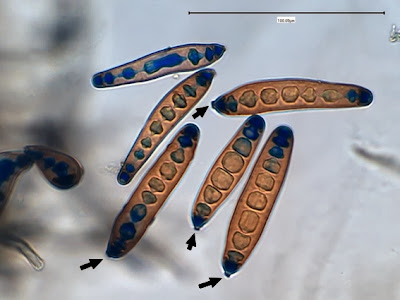
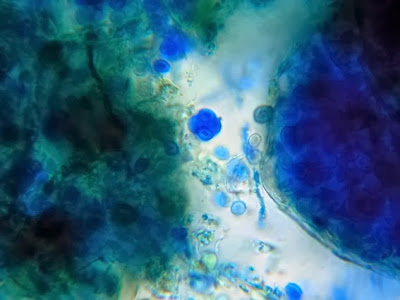
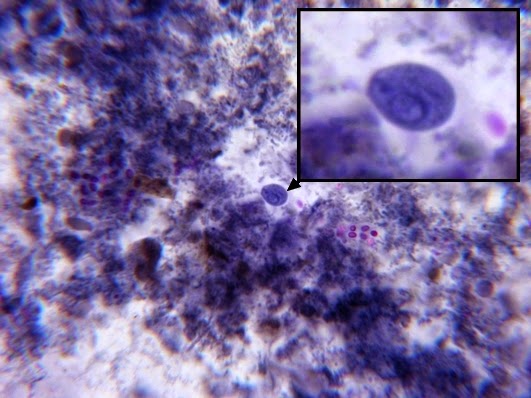
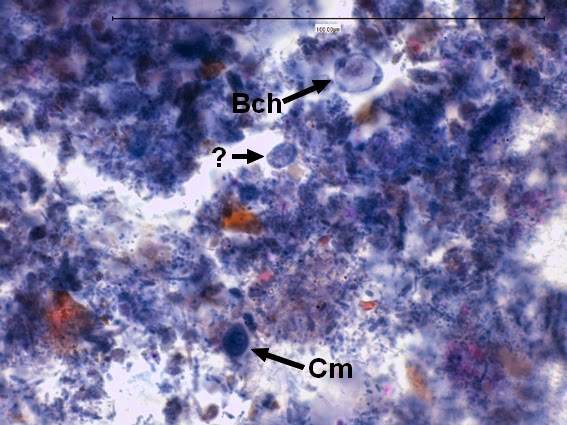
Helicobacter pylori - Compare the organism in the previous two photographs to Helicobacter pylori in this photo. The arrow in the inset photo points to just one H.pylori organism showing the curved "S" shape or "gull-wing" shape of the bacillus. Look carefully, the entire photo is teaming with H.pylori cells.
(Nikon, Carbol Fuchsin Stain, X1000)
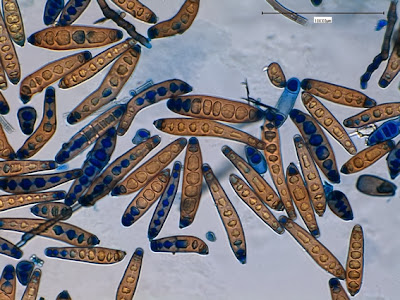
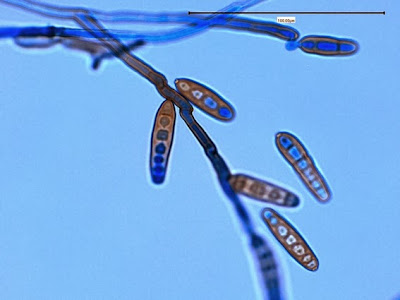
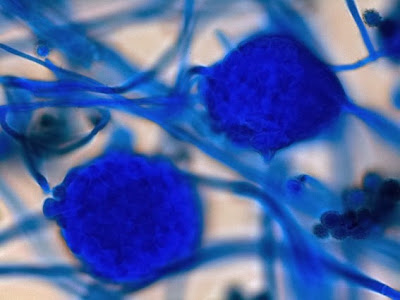
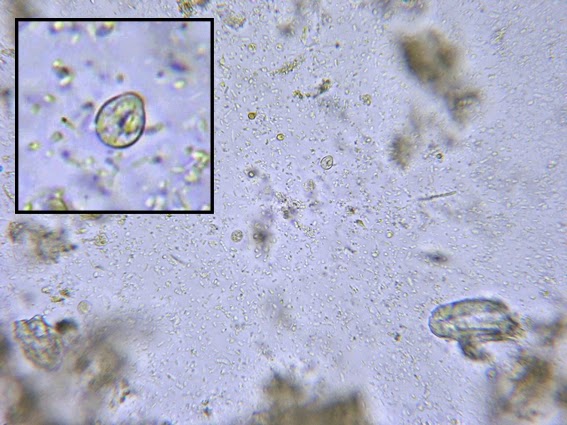
Campylobacter jejuni - Compare the Helicobacter species in the previous photographs to Campylobacter sp in the one immediately above. Campylobacter also exhibits a curved or spiral (helical) morphology. In fact, Helicobacter pylori was initially called Campylobacter pylori. Here we can see a greater number of twists or spirals but there are not as many, nor are they as tightly wound as with H.heilmanii. While C.jejuni is usually isolated from fecal specimens, this photo shows the organism in a blood culture.
(Nikon, Carbol Fuchsin Stain, X1000)
To read more about Campylobacter pylori, check out my Blog post specific to that organism by clicking here to redirect.
[i]Lancet 1987; 2:96
Dent, JC, McNulty CAM, Uff JC. Wilkinson, SP Gear MWL.
Spiral organisms in the gastric antrum.
[ii]Am J Gastroenterol. 1997 Aug;92(8):1310-5
Prospective, multivariate evaluation of CLOtest performance.
Weston AP, Campbell DR, Hassanein RS, Cherian R, Dixon A, McGregor DH.
Department of Veterans Affairs Medical Center, Kansas City, Missouri 64128-2226, USA
[iii]Rev Esp Enferm Dig. 1996 Mar;88(3):202-8.
C13 urea breath test in the diagnosis of Helicobacter pylori infection in the gastric mucosa. Validation of the method.
Pérez García JI, Pajares García JM, Jiménez Alonso I.
[iv]Eur J Gastroenterol Hepatol. 1999 Nov;11(11):1251-4.
The CLO test in the UK: inappropriate reading and missed results.
Prince MI, Osborne JS, Ingoe L, Jones DE, Cobden I, Barton JR.
University of Newcastle Regional School of Medicine, North Tyneside Hospital, North Shields, U
The CLO test in the UK: inappropriate reading and missed results.
Prince MI, Osborne JS, Ingoe L, Jones DE, Cobden I, Barton JR.
University of Newcastle Regional School of Medicine, North Tyneside Hospital, North Shields, U
v Practical Gastroenterology, February 2006; 47-50
Clinical Significance of Helicobacter heilmanii Colonizing Human Gastric Antrum
Anup Hazra, Carlos Ricart and Januz J. Godyn
Dept of Pathology & Laboratory Medicine & Dentistry, New Jersey; Robert Wood Johnson Medical School, New Brunswick, New Jersey
Clinical Significance of Helicobacter heilmanii Colonizing Human Gastric Antrum
Anup Hazra, Carlos Ricart and Januz J. Godyn
Dept of Pathology & Laboratory Medicine & Dentistry, New Jersey; Robert Wood Johnson Medical School, New Brunswick, New Jersey
vi Tzu Chi Med J, 2004; 16: No1 59-62
Helicobacter heilmanii of the Stomach – A Case Report
Jeh-En Tzeng, Ying-Lung Lin, Yi-Tsui Chu, Sue-Mei Chung
Department of Pathology, Family Medicine, Buddhist Dalin Tzu Chi General Hospital, Chiayi, Taiwan
Helicobacter heilmanii of the Stomach – A Case Report
Jeh-En Tzeng, Ying-Lung Lin, Yi-Tsui Chu, Sue-Mei Chung
Department of Pathology, Family Medicine, Buddhist Dalin Tzu Chi General Hospital, Chiayi, Taiwan
* * *
.jpg)



















.jpg)
.jpg)
+14d.jpg)









.jpg)
.jpg)
.jpg)
.jpg)














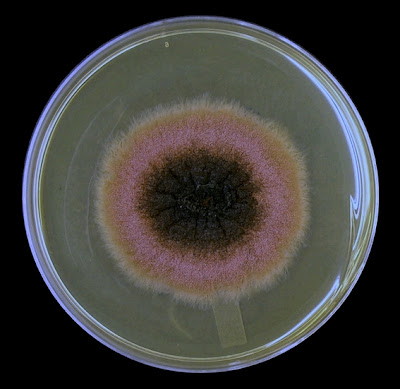+5+days.jpg)
+14+days.jpg)
.jpg)
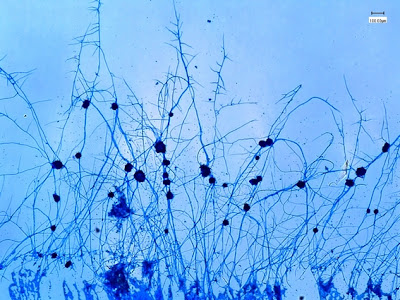
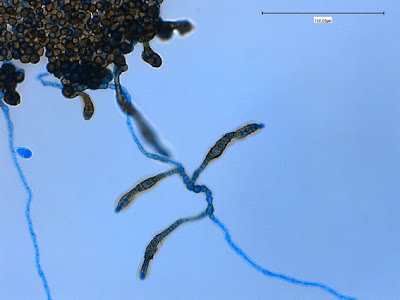

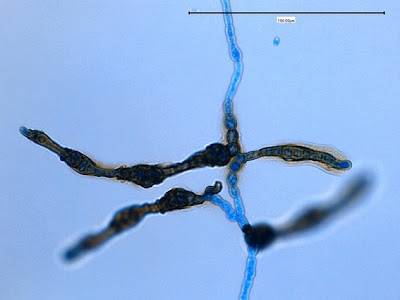








.jpg)






































.jpg)
.jpg)
.jpg)
+Reverse.jpg)
.jpg)
.jpg)

























.jpg)
.jpg)


















.jpg)



.jpg)
.jpg)


.jpg)













+72Hrs.jpg)
+72Hrs.jpg)






















































.jpg)